In 1822, French physicist Charles Cagniard de la Tour put a liquid and a flint ball into a Papin pressure vessel constructed using a rifle and heated the liquid in a sealed cannon. He then conducted an experiment to determine whether a product was produced.1) When the container was shaken, a splashing sound was heard as the ball penetrated the liquid-gas interface. However, when the container was heated to a temperature sufficiently higher than the boiling point of the liquid, the sound disappeared. He postulated that this was because the liquid and gas densities in the container became equal, so that a single phase was effectively formed. This represented the first discovery of what are now referred to as supercritical states and critical points.
Following this discovery, supercritical fluids became the subject of fundamental physicochemical research to determine the change in the state of materials and their physical properties under supercritical conditions. However, no practical applications immediately emerged. Then, in 1879, Hannay and Hogarth reported that supercritical fluids possessed excellent solvent properties,2) and a second wave of research began.
Substances can be in a solid, liquid, or gaseous state depending on the temperature and pressure conditions. For example, water is a liquid at room temperature and pressure, but at atmospheric pressure it changes to vapor (gas) at 100 degree C and ice (solid) at 0 degree C.
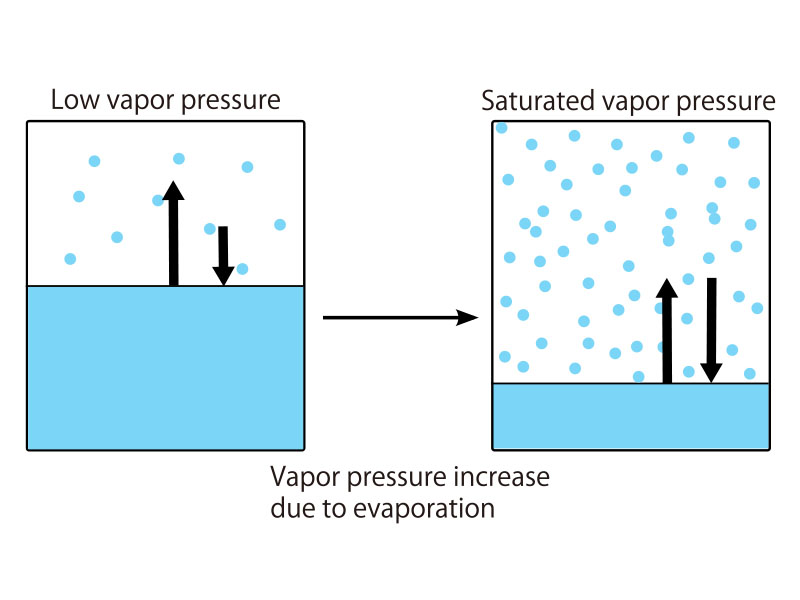
When water is placed inside a sealed container and subjected to a vacuum as shown in Figure 1, some of the water evaporates and the remainder remains in a liquid phase. When the water vapor pressure reaches a certain value, the evaporation rate becomes equal to the condensation rate. This is referred to as the saturated water vapor pressure, and depends on temperature.
As the vessel is heated, the liquid water expands and evaporates, so that its density is reduced. This leads to an increase in the density of the vapor phase. If the temperature exceeds 374 degree C and the pressure exceeds 22.06 MPa, the density of the liquid and vapor phases become the same, making it impossible to distinguish between liquid water and water vapor.
The water in this state will not become liquid even if the pressure is increased further. In other words, it’s like a dense gas that doesn’t liquefy.
Such a state is called a supercritical state, and a substance in a supercritical state is called a supercritical fluid. The temperature and pressure at which a substance becomes supercritical is called the critical point, the temperature is called the critical temperature, and the pressure is called the critical pressure.
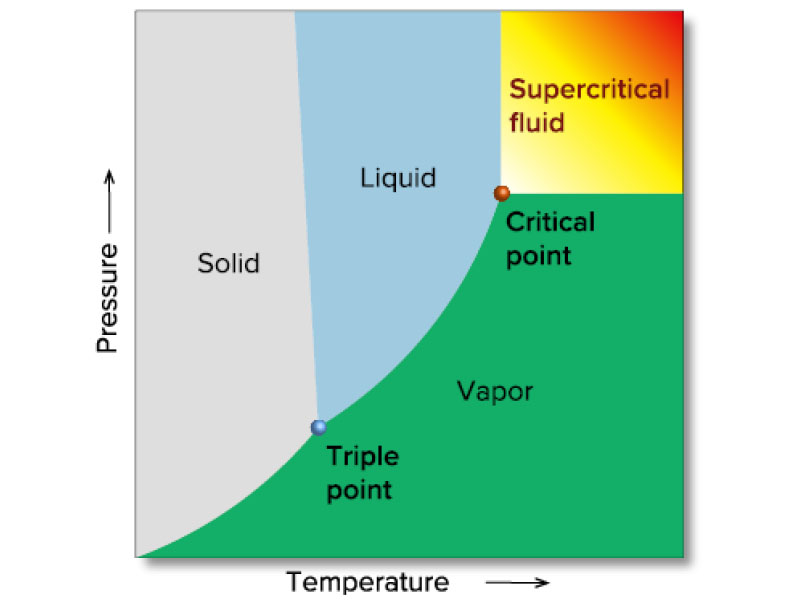
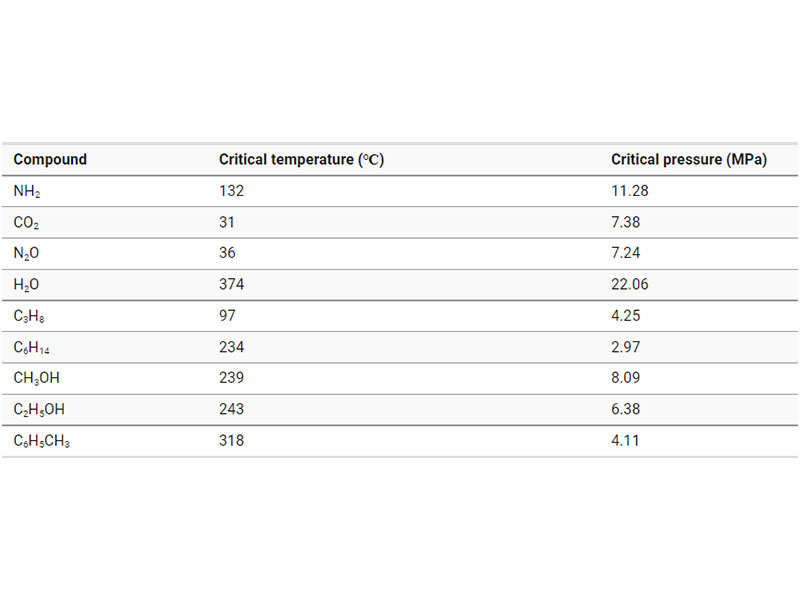
Figure 3 shows a phase diagram for an arbitrary substance, and Table 1 shows the critical temperature and critical pressure for various real compounds.
The density of a supercritical fluid is close to that of a liquid, and such a fluid is a highly effective solvent. However, the viscosity of a supercritical fluid is close to that of a gas, and the diffusion coefficient of a substance in a supercritical fluid is about halfway between that in a liquid and in a gas. In general, supercritical fluids exhibit properties that are intermediate between those of liquids and gases, and these properties are greatly influenced by temperature and pressure. As a result, supercritical fluids are used in a variety of applications such as mobile phases in chromatography, extraction agents, and chemical reaction solvents.
All substances are in a supercritical state above the critical point, but those with a high critical pressure and critical temperature are not practical. Carbon dioxide is a commonly used supercritical fluid, since its critical temperature is just 31.1 degree C and its critical pressure is only 7.38 MPa.
It offers the following advantages:
Supercritical fluid extraction (SFE) is a method in which supercritical fluid as an extraction medium is added to substances containing target components, and extraction is performed based on differences in solubility. In particular, the use of supercritical carbon dioxide as an extraction medium offers many advantages in a variety of fields, including short extraction times, simplified operation, and improved extraction efficiency compared with the organic solvent extraction method. Also, solvent removal following extraction is much easier, allowing more accurate component concentrations to be determined.
Because the critical temperature of carbon dioxide is only 31 °C, extraction is possible close to room temperature. Furthermore, extraction can be carried out in a carbon dioxide atmosphere without oxygen. This means that this method can be used for substances that are temperature-sensitive or easily oxidized. Supercritical carbon dioxide is effective at dissolving lipid-soluble or low-polar compounds, but it can be adapted to the extraction of highly polar components by adding alcohols such as methanol or organic solvents such as acetonitrile and tetrahydrofuran as auxiliary solvents (referred to as entrainers).
In recent years, it has attracted attention as an environmentally friendly extraction method that does not use harmful organic solvents, in line with the concept of Green Chemistry.
©Jasco Europe S.R.L. (Socio Unico) Direzione e coordinamento ex. art. 2497 bis c.c. – P.I. 08609570158 | Privacy Policy | Cookie Policy | Manage consent | Realizzazione sito web: Alkimedia
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| CookieLawInfoConsent | 1 year | Records the default button state of the corresponding category & the status of CCPA. It works only in coordination with the primary cookie. |
| elementor | never | This cookie is used by the website's WordPress theme. It allows the website owner to implement or change the website's content in real-time. |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |
| Cookie | Duration | Description |
|---|---|---|
| Google Maps | Google Maps is a map visualization service managed by Google Inc. and is used to integrate such contents within its pages. |