The demands of therapeutic antibody are increasing day by day, and CD spectroscopy attracts attention as the tool for assessing the higher-order structure of proteins quickly and easily.
The international council for harmonization ICH-Q6B guideline recommends CD spectroscopy as one of the methods for physicochemical characterization of biopharmaceuticals, and there is the requirement for an objective assessment of structural similarity described in ICH-Q5E. The FDA’s guidance places the most important teir1 method on objective and statistical evaluation of analytical similarity between biosimilars and their innovators. There is a growing demand for a process to objectively determine the similarity of CD spectra.
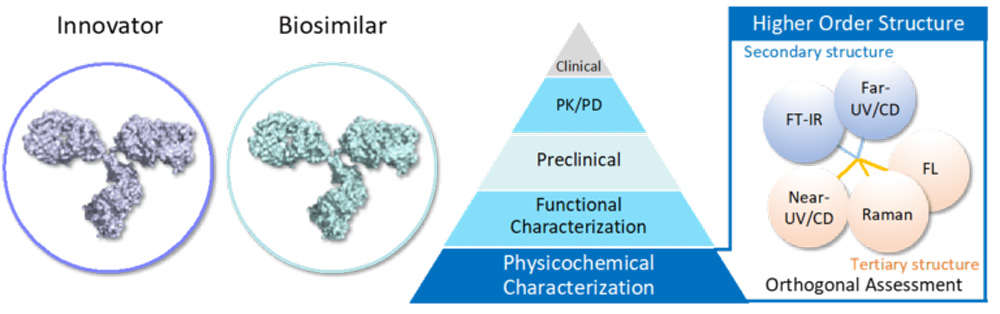
qHOS is a software that enables the statistical determination of spectral agreement, which is conventionally done by visual inspection. Weighting that takes into account spectral noise and weighting by external stimuli can be used to detect small differences in spectra with high sensitivity and robustness.
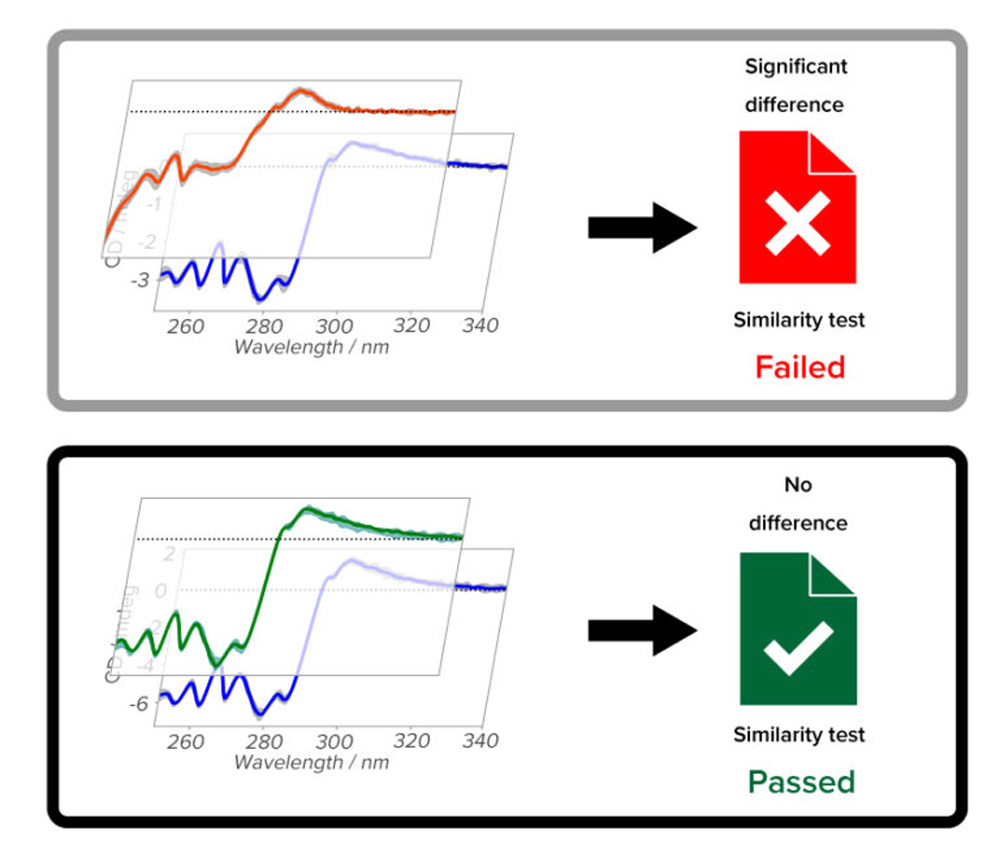
Spectroscopic data from various JASCO spectroscopic instruments can be analyzed on a single platform, supporting the orthogonal assessment required in the biotherapeutic development and quality control.